Published 1 September 2023 in Neuron (doi 10.1016/j.neuron.2023.08.006):
Structure and dynamics of differential ligand binding in the human ρ-type GABAA receptor
John Cowgill*, Chen Fan*, Nandan Haloi, Yuxuan Zhuang, Rebecca J Howard°, Erik Lindahl°
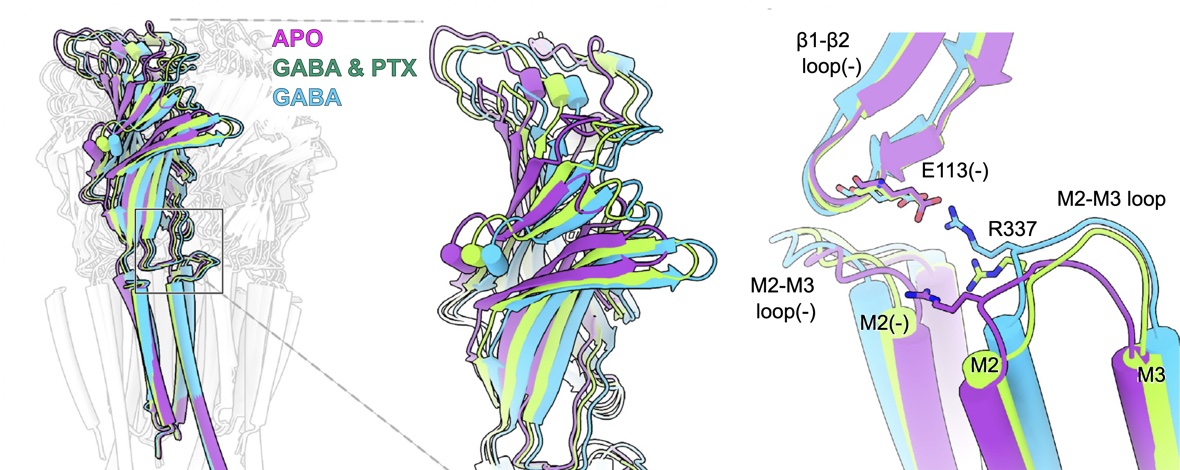
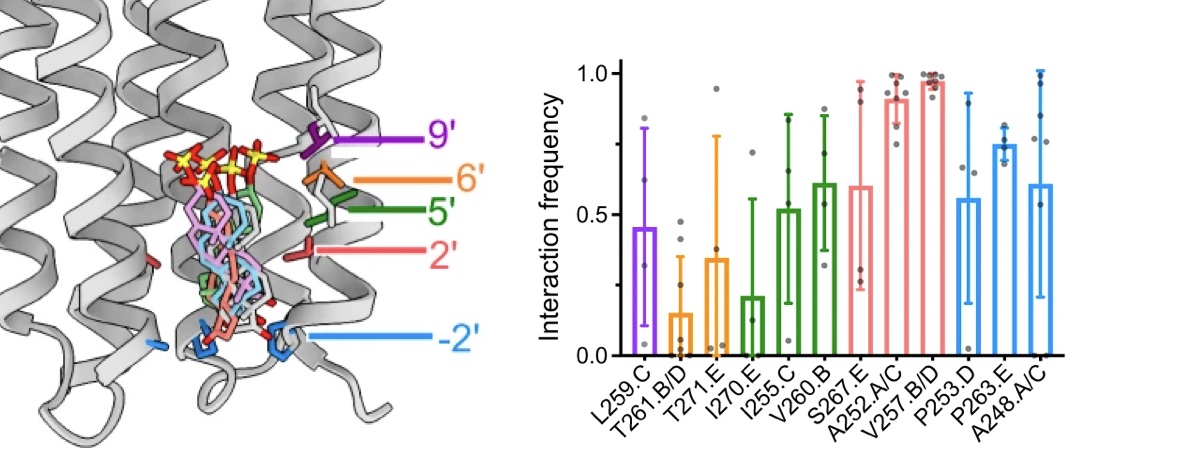
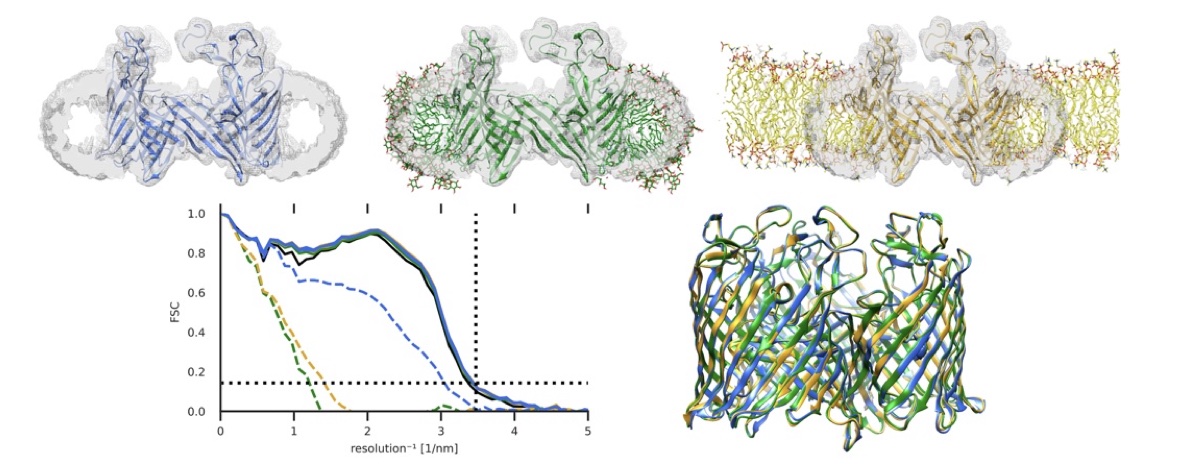
The neurotransmitter γ-aminobutyric acid (GABA) drives critical inhibitory processes in and beyond the nervous system, partly via ionotropic type-A receptors (GABAARs). Pharmacological properties of ρ-type GABAARs are particularly distinctive, yet the structural basis for their specialization remains unclear. Here, we present cryo-EM structures of a lipid-embedded human ρ1 GABAAR, including a partial intracellular domain, under apo, inhibited, and desensitized conditions. An apparent resting state, determined first in the absence of modulators, was recapitulated with the specific inhibitor (1,2,5,6-tetrahydropyridin-4-yl)methylphosphinic acid and blocker picrotoxin and provided a rationale for bicuculline insensitivity. Comparative structures, mutant recordings, and molecular simulations with and without GABA further explained the sensitized but slower activation of ρ1 relative to canonical subtypes. Combining GABA with picrotoxin also captured an apparent uncoupled intermediate state. This work reveals structural mechanisms of gating and modulation with applications to ρ-specific pharmaceutical design and to our biophysical understanding of ligand-gated ion channels.
*Contributed equally
°Corresponding authors
Read the full publication here.