Featured on the April 24, 2018 cover of Cell Reports (v. 23 pp. 993–1004):
Structural basis for a bimodal allosteric mechanism of general anesthetic modulation in pentameric ligand-gated ion channels
Zaineb Fourati*, Rebecca J Howard*, Stephanie A Heusser, Haidai Hu, Reinis R Ruza, Ludovic Sauguet, Erik Lindahl** & Marc Delarue**
*Equal contributions; **senior authors
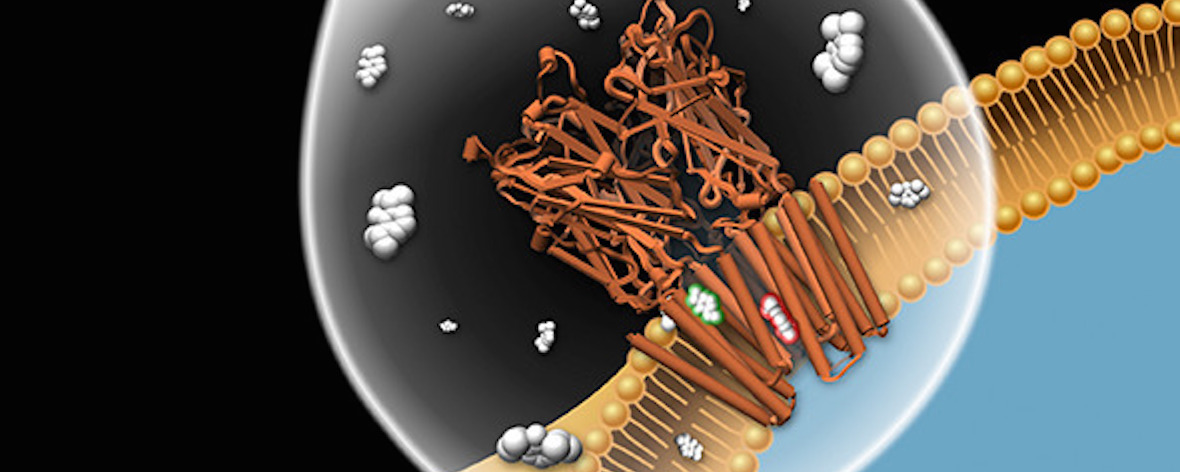
Ion channel modulation by general anesthetics is a vital pharmacological process with implications for receptor biophysics and drug development. Functional studies have implicated conserved sites of both potentiation and inhibition in pentameric ligand-gated ion channels, but a detailed structural mechanism for these bimodal effects is lacking. The prokaryotic model protein GLIC recapitulates anesthetic modulation of human ion channels, and it is accessible to structure determination in both apparent open and closed states. Here, we report ten X-ray structures and electrophysiological characterization of GLIC variants in the presence and absence of general anesthetics, including the surgical agent propofol. We show that general anesthetics can allosterically favor closed channels by binding in the pore or favor open channels via various subsites in the transmembrane domain. Our results support an integrated, multi-site mechanism for allosteric modulation, and they provide atomic details of both potentiation and inhibition by one of the most common general anesthetics.
Read the full publication here.