Published online 31 July 2023 in PLOS Computational Biology (doi 10.1371/journal.pcbi.1011255):
Gentle and fast all-atom model refinement to cryo-EM densities via a maximum likelihood approach
Christian Blau, Linnea Yvonnesdotter, Erik Lindahl
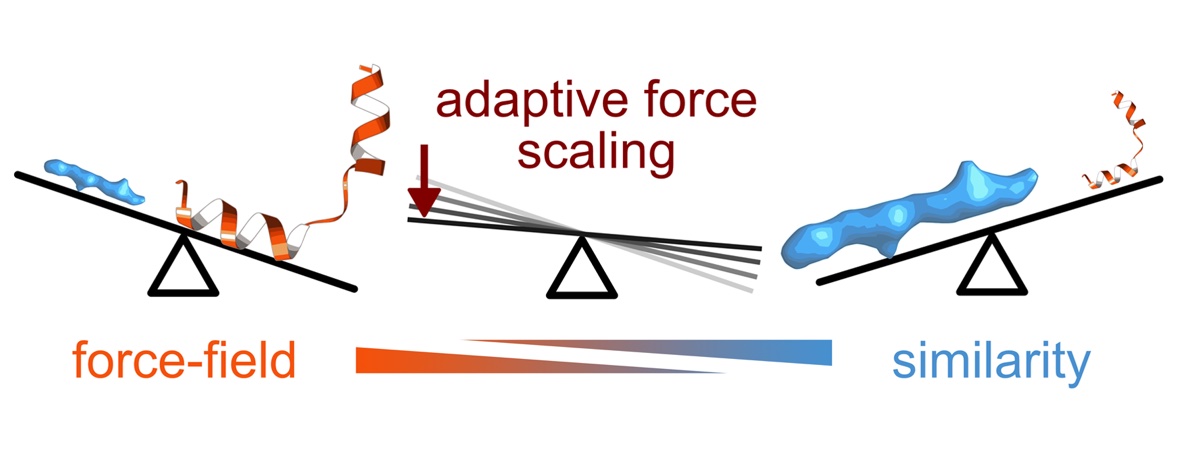
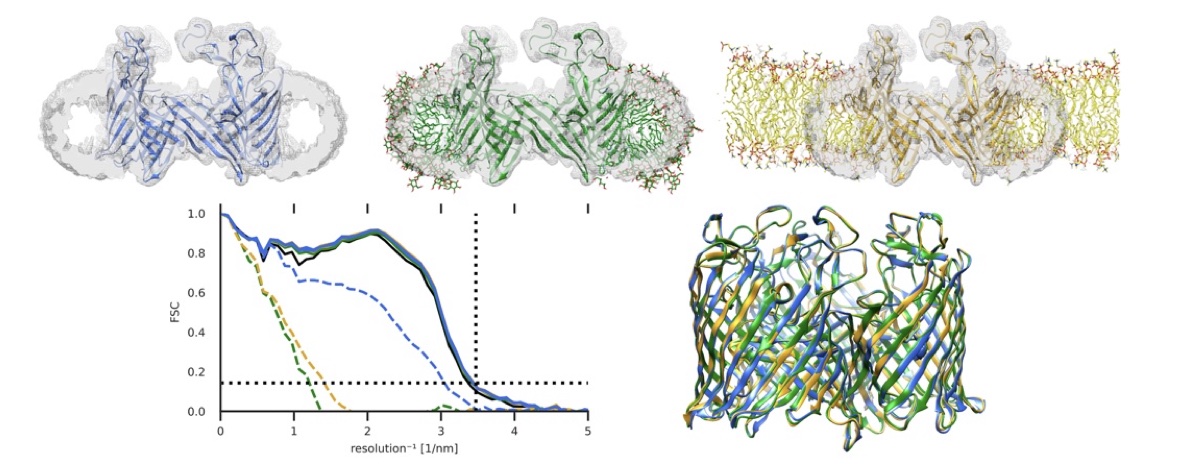
Better detectors and automated data collection have generated a flood of high-resolution cryo-EM maps, which in turn has renewed interest in improving methods for determining structure models corresponding to these maps. However, automatically fitting atoms to densities becomes difficult as their resolution increases and the refinement potential has a vast number of local minima. In practice, the problem becomes even more complex when one also wants to achieve a balance between a good fit of atom positions to the map, while also establishing good stereochemistry or allowing protein secondary structure to change during fitting. Here, we present a solution to this challenge using a maximum likelihood approach by formulating the problem as identifying the structure most likely to have produced the observed density map. This allows us to derive new types of smooth refinement potential—based on relative entropy—in combination with a novel adaptive force scaling algorithm to allow balancing of force-field and density-based potentials. In a low-noise scenario, as expected from modern cryo-EM data, the relative-entropy based refinement potential outperforms alternatives, and the adaptive force scaling appears to aid all existing refinement potentials. The method is available as a component in the GROMACS molecular simulation toolkit.
Read the full publication here.