For the October 1, 2018 issue of the Journal of General Physiology (v. 150 art. 1444):
Determining the molecular basis of voltage sensitivity in membrane proteins
Marina A. Kasimova, Erik Lindahl & Lucie Delemotte
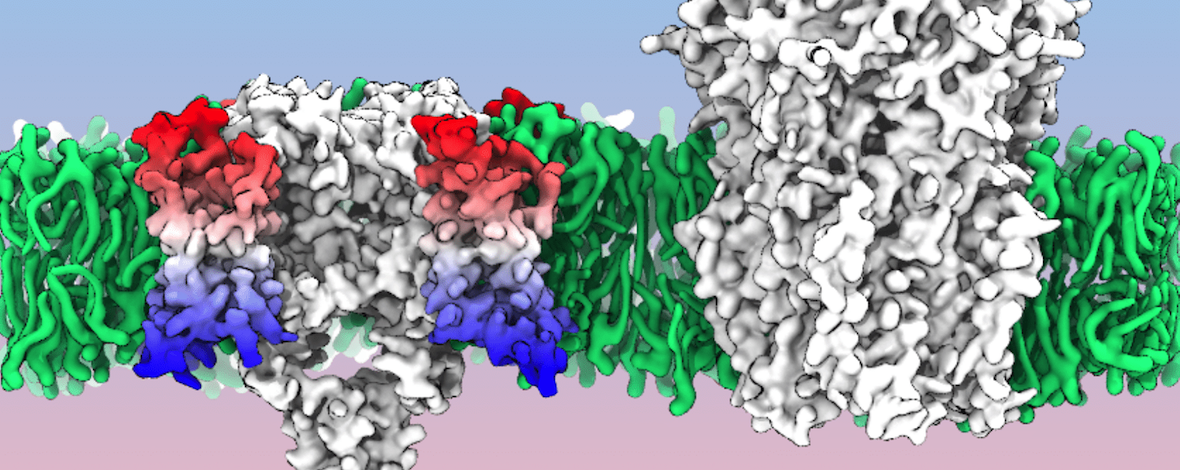
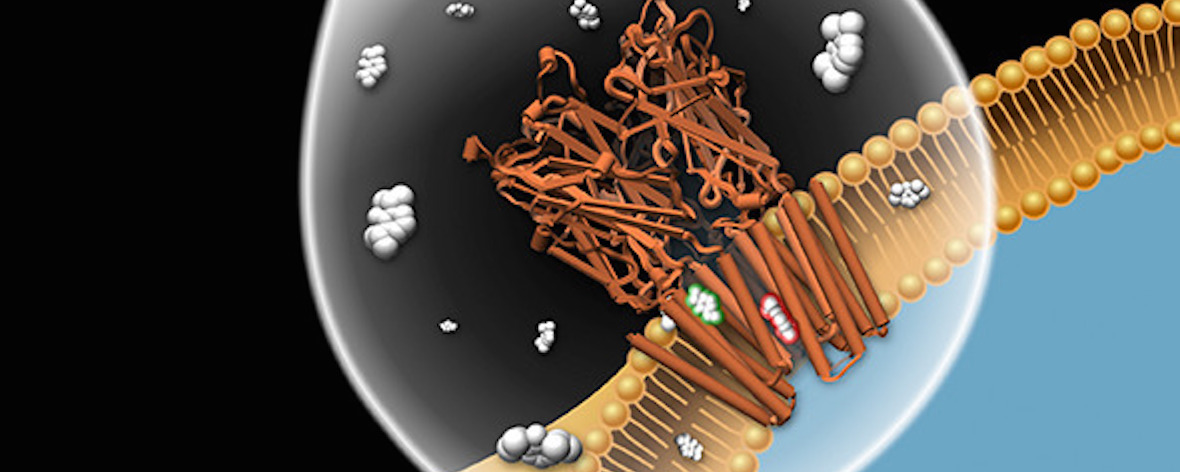
Voltage-sensitive membrane proteins are united by their ability to transform changes in membrane potential into mechanical work. They are responsible for a spectrum of physiological processes in living organisms, including electrical signaling and cell-cycle progression. Although the mechanism of voltage-sensing has been well characterized for some membrane proteins, including voltage-gated ion channels, even the location of the voltage-sensing elements remains unknown for others. Moreover, the detection of these elements by using experimental techniques is challenging because of the diversity of membrane proteins. Here, we provide a computational approach to predict voltage-sensing elements in any membrane protein, independent of its structure or function. It relies on an estimation of the propensity of a protein to respond to changes in membrane potential. We first show that this property correlates well with voltage sensitivity by applying our approach to a set of voltage-sensitive and voltage-insensitive membrane proteins. We further show that it correctly identifies authentic voltage-sensitive residues in the voltage-sensor domain of voltage-gated ion channels. Finally, we investigate six membrane proteins for which the voltage-sensing elements have not yet been characterized and identify residues and ions that might be involved in the response to voltage. The suggested approach is fast and simple and enables a characterization of voltage sensitivity that goes beyond mere identification of charges. We anticipate that its application before mutagenesis experiments will significantly reduce the number of potential voltage-sensitive elements to be tested.
Read the commentary in the same issue by Caitlin Sedwick, or see the paper here!